Quantum mechanics is the study of how very, very small particles (electrons, atoms, light, etc) act. Think of a bouncy ball. If the ball is a normal size you know exactly where it is and how much energy it has (you know how it will bounce). The normal size bouncy ball follows classical mechanics.
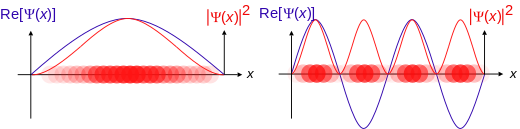
Now imagine that ball were the size of an atom. Just by being this size, the ball will follow completely different laws of physics known as quantum mechanics. The ball will now act like in this diagram. Notice that you can't say exactly where the ball is. You also don't know exactly how it will act. Instead it is spread out over a large area. You can know where the ball probably is, and you can know how it would probably act. The new properties of this ball are described by a wavefunction. The wavefunction is a mathematical statement that describes the energy of the ball.
Now, notice that this ball has properties that sort of look like a wave. Very small particles act like a wave. This is known as wave-particle duality - the ball acts like a wave and a particle. When we aren't looking at the ball it is acting like a wave. It is everywhere at the same time, with higher probability in some places than others. However, the simple act of looking1 at the ball makes it act like a normal ball again. When the ball changes from being everywhere at once to only in one place we say that the wavefunction has collapsed.
So, why did I call my blog "The Collapsed Wavefunction"? Well, partly because I'm a nerd and wanted the name to be something nerdy. But there is a real reason for choosing the name. My goal is to write about science topics in a way that everyone can understand. Many of you may see these topics like the quantum ball - you know something about it, but your understanding is a bit blurry. By looking at subjects you might be unsure about, we are collapsing the wavefunction, and that's my goal for this blog.
Notes
[1] Those that have studied quantum mechanics may want to point out that looking at it is not quite true that looking at a particle is what collapses the wavefunction. The actual term is observe, and has nothing to do with seeing anything at all. An observer doesn't even have to be something living. Light is a good example of an observer. When light strikes an atom, and we follow what happens, the wavefunction of the atom is collapsed and we can know what state the atom was in.