...You're still here. Ok, good.
Basically, the first law says that energy can't be created or destroyed. Instead, energy can be converted from one form to another. For example, if I'm holding a ball high in the air it has potential energy. Energy is stored in the ball because of its height. If I let that ball go the stored energy is converted to kinetic energy and it falls to the ground. That's the example you'll see in about every textbook - here's a more real world explanation:
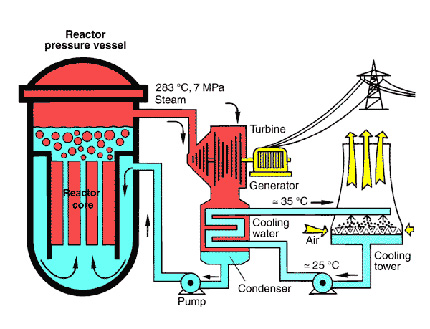
A power plant (nuclear, wind, water, coal, solar - any type of power) doesn't generate energy - that would break the first law. Instead, energy is converted from one form to another. Nuclear power plants generate electricity by nuclear fission - a reaction that releases (not creates) large amounts of energy. This energy heats a bath of water and creates steam. The steam then turns a turbine that generates electricity[1]. All other forms of harnessing energy work in similar ways (wind spins a turbine, coal creates steam that turns a turbine etc).
Sometimes will they have created a that shows thermodynamics is wrong. Mostly they just don't understand science or sometimes they don't realize where energy is being lost or coming from. Watch the videos I linked. They're pretty funny in some cases. Here's one thing to look for - if the machine makes noise it's loosing energy somewhere. You need to realize that if these people were creating energy they would not only be world famous but filthy rich. Energy is a global issue and if a solution were that readily available it would be exploited - the first law is not a conspiracy.
The first law is stated simply enough, but still misunderstood (or ignored) by self-titled geniuses who are certain they have solved the energy crisis. The reality, though, is that the first law is in our favor not working against us.
Notes
[1] The large cylinders that most people associate with a nuclear power plant is the cooling tower, seen above. It's basically just an outlet for steam. Neither radiation nor radioactive material escape from them (The difference between radiation and radioactive material may be discussed later).
